Competency 3 – Conceptual and quantitative knowledge of energy and its effects
1. Differentiate between forms of energy and their transformations.
- Energy – an abstract property defined as the capacity to do work; there are a number of forms of energy, but most of them can be reduced to either kinetic energy or potential energy
- Kinetic energy – the energy an object possesses by virtue of its motion; KE = ½ mv2
- Radiant energy – electromagnetic energy that travels in transverse waves
- Includes visible light, x-rays, gamma rays, and radio waves
- Thermal energy – the energy that comes from the movement of atoms and molecules in a substance; heat
- Sound energy – energy moving through substances in longitudinal waves
- Sound is produced when a force causes an object or substance to vibrate
- Electrical energy – delivered by electrons, typically moving through a wire
- Radiant energy – electromagnetic energy that travels in transverse waves
- Potential energy – the energy an object possesses by virtue of its position relative to others, stresses within itself, electric charge, and other factors
- Gravitational potential energy – the energy stored within an object as the result of its vertical position or height; the energy is stored as a result of the gravitational attraction of the Earth; PEgrav = mgh
- Elastic potential energy – the energy stored in elastic materials as the result of their stretching and compressing; PEspring = ½ kx2
- Chemical energy – energy stored in the bods of atoms and molecules
- Nuclear energy – energy stored in the nucleus of an atom; can be released when the nuclei are combined or split apart
- Mechanical energy – the energy acquired by an object upon which work is done
- Can be either kinetic energy or potential energy
- TME = PE + KE
- Kinetic energy – the energy an object possesses by virtue of its motion; KE = ½ mv2
- Thermodynamics – the study of how energy changes from one type to another
- First Law of Thermodynamics – energy cannot be created or destroyed, however it can be transformed from one type to another
- Energy transformations – processes that convert energy from one type into another; any type of energy use must involve some sort of energy transformation
- Rubbing hands together to make them warm: kinetic energy → thermal energy
- Using a battery powered flashlight: chemical energy → electrical energy → radiant energy
- An object speeding up as it falls: gravitational potential energy → kinetic energy
- Energy transformations – processes that convert energy from one type into another; any type of energy use must involve some sort of energy transformation
- Second Law of Thermodynamics – some forms of energy are more useful than others; using energy always makes it less useful, even though no energy is actually destroyed
- Most useful: kinetic energy and electricity
- Least useful: thermal energy
- First Law of Thermodynamics – energy cannot be created or destroyed, however it can be transformed from one type to another
2. Relate energy to transitions between states of matter.
- Transitions involving an increase in energy – endothermic phase changes
- More ordered state → less ordered state
- Heat absorbed causes the molecules to move farther apart by overcoming the intermolecular forces of attraction
- Matter + thermal energy → endothermic phase change (kinetic energy ↑)
- Melting – solid → liquid
- Evaporation – liquid → gas
- Sublimation – solid → gas
- Transitions involving a decrease in energy – exothermic phase changes
- Less ordered state → more ordered state
- Heat released allows the molecules to move closer together and the intermolecular forces of attraction become stronger
- Matter – thermal energy → exothermic phase change (kinetic energy ↓)
- Deposition – gas → solid
- Condensation – gas → liquid
- Freezing – liquid → solid
3. Distinguish between temperature, heat, and thermal energy.
- Temperature – the average kinetic energy of molecules within a system or material; measured in Celsius (℃), Fahrenheit (℉), or Kelvin (K)
- Heat – the transfer of thermal energy between molecules within a system; measures how energy moves or flows (Joules)
- Thermal energy – energy contained in the motion and vibration of its molecules
4. Distinguish between the types of thermal energy transfer (e.g., radiation, conduction, convection).
- Radiation – does not necessarily require a medium to transfer it; facilitated through a type of electromagnetic radiation
- Emitted electromagnetic waves will move until they hit another particle
- The particle receives this radiation as kinetic energy
- How Earth gets energy from the sun
- Conduction – the movement of kinetic energy in materials from higher temperature to lower temperature through a substance
- Molecules will give their energy to adjacent molecules until equilibrium is reached
- Does not deal with the movement of particles within the material
- Transfer of heat through direct contact, such as on a stove
- Convection – heat transfer through fluid motion
- Motion usually occurs due to differences in density
- Higher temperature particles → cooler areas
- Lower temperature particles → warmer areas
- The fluid will remain in motion until equilibrium is reached
- The cooling of a room due to currents in the air
- The cooling of a room due to currents in the air
5. Apply the laws of thermodynamics to real-world situations.
- Melting of ice in a beverage
- A few hours after placing an ice cube in a warm beverage, the ice has melted and the beverage has cooled
- The total amount of heat in the system has remained the same but has gravitated toward equilibrium
- The melted ice and the beverage are now the same temperature
- The beverage will eventually become warm again as heat from the environment is transferred into the glass and its contents
- Sweating in a crowded room
- While in a small, crowded room with lots of other people, you will start to feel warm and begin sweating
- Heat from your body is transferred to the sweat
- Sweat evaporates from your body as it absorbs more heat
- Heat from the sweat is transferred to the surrounding air, increasing the air temperature of the room
- No heat is lost, it is merely transferred and approaches equilibrium with maximum entropy
- Taking a very long bath
- Initially the temperature of the water is very hot and feels comfortably warm, as it is a higher temperature than that of the body
- As time passes, some heat from the water is transferred to the person’s body and the two temperatures will meet
- As more time passes, the water will cool as heat is lost to the atmosphere
- The person will also cool, but not to the same extent as the water
- Charging a cell phone
- Water is slowly released through a small opening in a dam and turns a turbine
- The turbine is used to generate electricity with the help of a generator
- The electricity (AC) is sent through high-voltage power lines
- A transformer lowers the voltage to a safer range
- An AC adapter is plugged into an outlet and the AC is converted to DC
- The battery is charged
6. Differentiate between potential and kinetic energy.
- Kinetic energy – the energy an object possesses by virtue of its motion
- An object with a velocity of 30 m/s has more kinetic energy than an object of the same mass with a velocity of 15 m/s
- Potential energy – the energy an object possesses by virtue of its position relative to others, stresses within itself, electric charge, and other factors
- An object at a height of 100 m has more potential energy than an object of the same mass at a height of 50 m
- Rollercoaster example:
- At the top of the first hill, PE = max, KE = min
- As the car goes down the hill, KE increases and PE decreases
- At the bottom of the first hill, PE = min, KE = max
- As the car goes up the next hill, PE increases and KE decreases
7. Identify characteristics of nuclear reactions.
- Nuclear reaction – a change in identity or characteristics of an atomic nucleus
- reacting particles (a heavy target nucleus and a light bombarding particle) and produces two new particles (a heavier product nucleus and a lighter ejected particle)
- Types of nuclear reactions:
- Fission – a heavy nucleus is “split” into two or more smaller nuclei
- Exothermic and start with nuclei heavier than iron
- Widely used to generate electrical power using uranium as a fuel
- Fusion – when two or more lighter nuclei come together to make a heavy nucleus
- The fusion of four hydrogen atoms and two electrons into a single helium atom is the primary reaction in the sun
- Nuclear decay – the process by which an unstable isotope of a particular element spontaneously transforms into a new element by emission of ionizing radiation; three common emissions occur:
- Alpha particles (α) – 42 α (mass number + 4; atomic number + 2)
- Penetrating power – very low
- Ionizing power – very high
- Shielding – paper skin
- Beta particles (β) – 0-1 β (mass number + 0; atomic number – 1)
- Penetrating power – intermediate
- Ionizing power – very low
- Shielding – two inches lead
- Gamma rays (γ) – 00 γ
- Penetrating power – very high
- Ionizing power – very high
- Shielding – paper skin
- Alpha particles (α) – 42 α (mass number + 4; atomic number + 2)
- Transmutation – the non-spontaneous process where one element is transformed into another by bombarding it with high energy radiation or neutrons
- Allows for the creation of radioactive isotopes
- Fission – a heavy nucleus is “split” into two or more smaller nuclei
8. Identify the regions of the electromagnetic spectrum and energy.
- The electromagnetic spectrum – the range of all types of electromagnetic radiation (energy that travels and spreads out as it goes); the full range of frequencies (from radio waves to gamma rays) that characterizes light
- Radio waves:
- Music on the radio
- emitted by stars in space
- can travel for a long distance, penetrate materials, cannot be felt, and travel at the speed of light in a vacuum
- Microwaves:
- Used in communications, radio astronomy, remote sensing, radar, and heating (cooking)
- Reflected by metal surfaces (why satellite dishes are made of metal)
- Certain frequencies are absorbed by water; water in food absorbs the microwaves which causes the water to heat up, therefore cooking the food
- Infrared waves:
- Can be detected as a sensation of warmth on the skin
- Most of the radiation emitted by a moderately heated surface
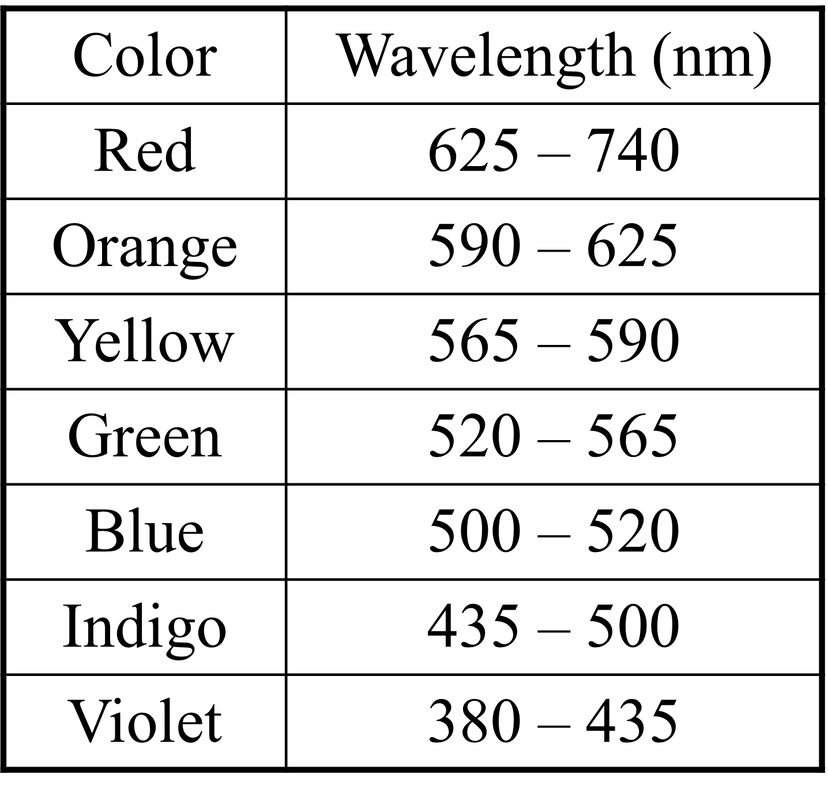
- Visible light – the segment of the electromagnetic spectrum that the human eye can view
- Most light we interact with is white light; shining white light through a prism causes the wavelengths to bed at slightly different angles due to optical refraction; the resulting light is split across the visible color spectrum
- Most light we interact with is white light; shining white light through a prism causes the wavelengths to bed at slightly different angles due to optical refraction; the resulting light is split across the visible color spectrum
- Radio waves:
- Ultraviolet rays:
- Can cause certain materials to fluoresce (glow; emit electromagnetic radiation of lower energy)
- Produced by high temperature surfaces (including the sun)
- Causes sunburn
- X-rays:
- A form of ionizing radiation; energetic enough to cause neutral atoms to eject electrons
- Can pass through skin and tissue but not bone
- Gamma rays:
- Produced in the disintegration of radioactive nuclei and in the decay of certain subatomic particles
- A form of ionizing radiation
- Used for PET scans and radiation therapies
9. Identify the use of light and optics in real-world applications (e.g., optical instruments, communication).
- Remote controls – contain a small laser that shoots across the room to the sensor on your TV, which also uses optics technology
- Cameras – photography uses light and shadow to create images
- 5G – uses fiber optic cables to transmit information, allowing radio signals to travel faster over longer distances and with less losses
- Barcode scanners – use lasers to read the pattern of lines and blank space on a barcode
- Traffic signals – use longer-lasting LED bulbs to be seen farther away
- Surveillance cameras – made it possible to record better video and make the technology more affordable
- LED light bulbs – last longer and use less energy than incandescent light bulbs
- Surgical machines – x-rays, MRI’s, PET scans, laser surgery, high powered microscopes
10. Solve problems involving energy, work, power, mechanical advantage, and efficiency.
- Energy – the capacity to do work
- Work – a process that occurs when a force acts over a distance in the same direction as the movement
- W = F × d; expressed in joules (J)
- Machines do not change the amount of work done, but they can reduce the force needed by increasing the distance
- Power – the rate at which work is done
- P = W/t; SI Unit: joules per second (J/s)
- Mechanical advantage – a measure of the ratio of output force to input force in a system
- MA = Fo/Fi; does not have a unit
- Efficiency – a way of describing the useful output a process or machine can generate as a percentage of the input required
- Efficiency = Eo/Ei × 100%
- Machine – any device that makes work easier by changing force
- Simple machines – a physical device that changes the magnitude or direction of a force; reduce the amount of force required to do work by increasing the distance; can use mechanical advantage to increase force
- Inclined plane – a sloping surface (ramp); offers a mechanical advantage in that the force required to move an object up the incline is less than the weight being raised
- Lever – a rigid bar or board that rests on a fulcrum; a downward force exerted on one end of the lever can be transferred and increased in an upward direction at the other end, allowing a small force to lift heavy weight
- Wedge – an object that tapers to a thin edge; pushing the wedge in one direction creates a force in a sideways direction; used for splitting, lifting, or tightening
- Wheel and axle – a circular frame (wheel) that revolves on a rod or shaft (axle)
- Pulley – a wheel that carries a flexible rope, cord, cable, chain, or belt on its rim; used singly or in combination to transmit energy and motion
- Screw – an inclined plane wrapped around a rod or shaft; used either as a fastener or as a force and motion modifier
- Compound machine – a machine that consists of more than one simple machine
- Simple machines – a physical device that changes the magnitude or direction of a force; reduce the amount of force required to do work by increasing the distance; can use mechanical advantage to increase force
11. Apply the laws of conservation of mass and energy to chemical reactions, nuclear reactions, physical processes, and biological processes.
- Chemical reactions:
- Conservation of mass: mass is neither created nor destroyed during chemical reactions
- The mass of any one element at the beginning of the reaction is equal to the mass of that element at the end of the reaction
- The total mass will be the same at any point during the reaction (in a closed system)
- Conservation of energy: energy may change form during a chemical reaction
- Conservation of mass: mass is neither created nor destroyed during chemical reactions
- Nuclear reactions:
- Conservation of mass: the law requires that during any nuclear reaction, radioactive decay, or chemical reaction in a closed system, the total mass of the reactants or starting materials must be equal to the mass of the products
- Conservation of energy: the total relativistic energy must be conserved
- Mass and energy are equivalent and convertible; E = mc2
- Physical processes:
- Conservation of mass: matter can change in form through a physical change, but the same amount of matter exists before and after the change
- Conservation of energy: the entire energy of the universe is conserved
- Endothermic: surroundings → system; heat is absorbed by the system and the surroundings get cooler
- Exothermic: system → surroundings; heat is released by the system and the surroundings get warmer
- Biological processes:
- Conservation of mass: living organisms are primarily made of six elements (oxygen, carbon, hydrogen, nitrogen, calcium, and phosphorous) and each of these elements cycle through the earth system
- Conservation of energy: energy is transformed from one form to another
- Photosynthesis: energy from the sun → absorbed by cells in plants → chemical energy (stored as glucose) → complex carbohydrates
- Cellular respiration: glucose + O2 → CO2 + H2O; allows plant or animal organisms to access the energy stored in carbohydrates, lipids and other macromolecules through the production of ATP
- The higher up an organism is on the food chain, the less available energy it receives from its food sources
12. Identify types, characteristics, and measurements of electrical quantities.
- Electrical current (Amperes, A) – the flow of charge; number of charges per unit time passing through a boundary
- Voltage (Volts, V) – electromotive force; the electric potential difference per unit charge between two points in an electric field (voltage = joule/coulomb)
- Electrical charge (Coulombs, C) – the property of subatomic particles that causes them to experience a force when placed in an electric and magnetic field; one coulomb is the quantity of charge transferred in one second
- Resistance (Ohms, Ω) – a force that counteracts the flow of current; how difficult it is for current to flow
- Capacitance (Farads, F) – the ability of an electric conductor to store an electrical charge
- Inductance (Henry, H) – property of a conductor that is measured by the voltage induced in it compared with the rate of change of the electric current that produces the voltage
- Electric power (Watts, W) – the rate energy is transferred or transformed over time
13. Apply knowledge of currents, circuits, conductors, insulators, and resistors to real-world situations.
- Circuit – a closed conducting loop in which a charge can flow
- Parts of a circuit –
- Electrical conductor (wire) – connects to the power source to form a closed loop
- Power source – source of electrical energy (such as a battery)
- Switch – a device to open and close a circuit
- Load – a device that the circuit is powering
- Capacitor – a device used to store an electric charge
- Resistor – a device that limits or regulates the flow of an electric current
- Types of circuits –
- Series circuit – a circuit where the components are connected end-to-end to form a single path for current to flow
- If one component breaks down, the whole circuit will burn out
- Parallel circuit – a circuit in which all components are connected across each other with exactly two electrically common nodes with the same voltage across each component
- Other components will function even if one component breaks down as each has its own independent circuit
- Series circuit – a circuit where the components are connected end-to-end to form a single path for current to flow
- Parts of a circuit –
- Conductor – materials that allow electrons to travel through
- Metals, aqueous solutions of salts, graphite, the human body
- Insulator – materials that inhibit the flow of electrons
- Plastics, Styrofoam, paper, rubber, glass, and dry air
- Plastics, Styrofoam, paper, rubber, glass, and dry air
14. Solve mathematical problems involving current, voltage, resistance, power, and energy in direct current (DC) circuits.
- Power Equation of Ohm’s Law and Joule’s Law
- Voltage (volts) – E
- Current (amperes) – I
- Resistance (ohms) – R
- Power (watts) – P
- Ohm’s Law – E = IR
- Joule’s Law – P = IE