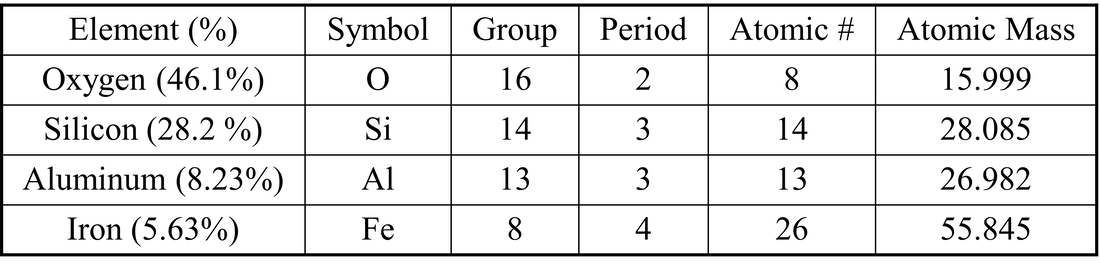
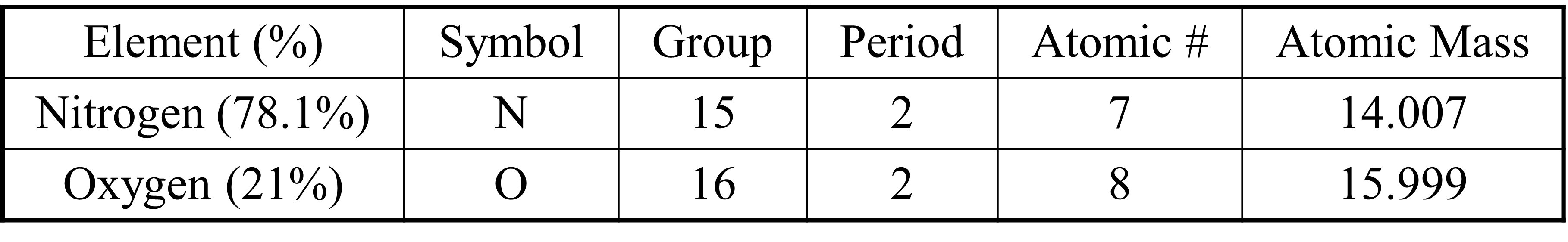
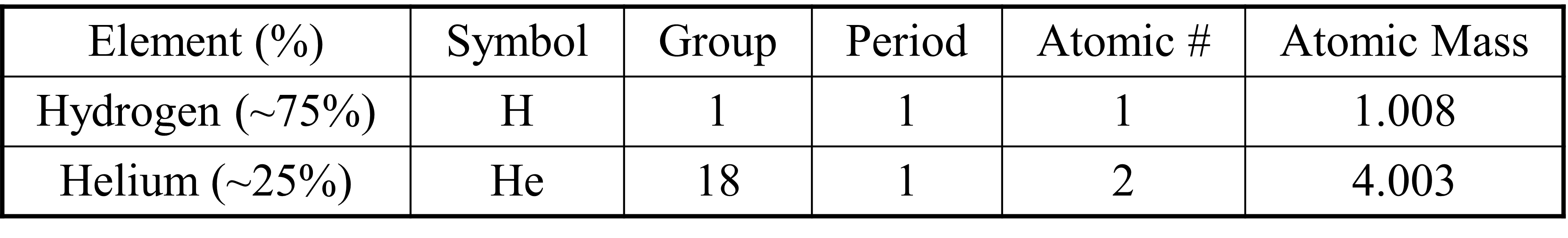
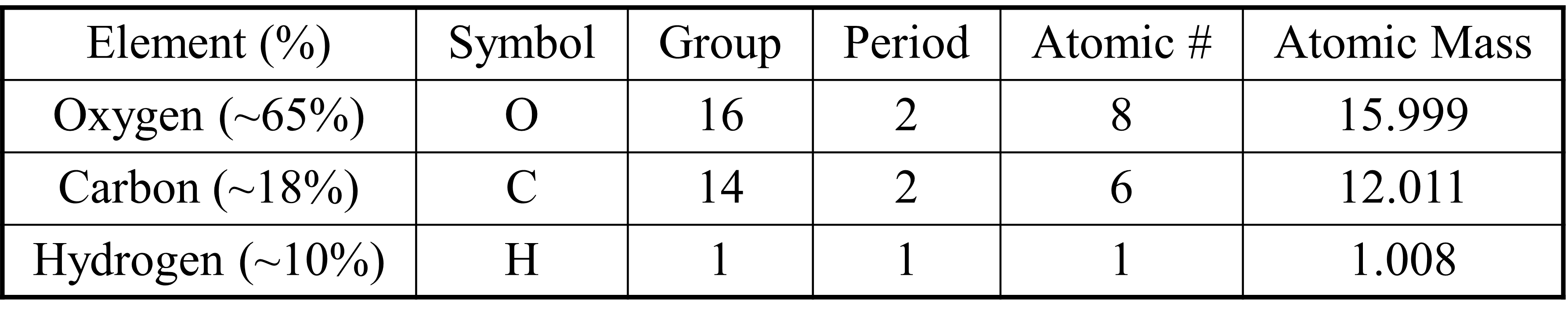
Competency 1 – Conceptual and quantitative knowledge of the structure and behavior of matter
1. Analyze the physical and chemical properties of matter (e.g. mass, volume, density, and chemical reactivity)
- Physical and chemical properties of matter
- Mass – the amount of matter that an object contains; different from weight as it is independent of the gravitational field exerted on the object; SI Unit = kilograms (kg)
- Volume – the amount of space taken up by matter; SI Unit = m3
- Density – a measure of the compactness of a substance given by the mass per unit volume; d=m/v; SI Unit = kg/m3
- Chemical reactivity – how readily a substance undergoes a chemical reaction; can involve the substance on its own or other atoms or compounds; generally accompanied by a release of energy; halogens, alkali metals, and alkaline earth metals are highly reactive; noble gases are the most stable elements
- Mass – the amount of matter that an object contains; different from weight as it is independent of the gravitational field exerted on the object; SI Unit = kilograms (kg)
2. Distinguish between states of matter
- States of matter:
- Solid – a collection of atoms or molecules that maintain a defined shape an size under constant conditions; can be crystalline or amorphous
- Liquid – a state of matter characterized by its condensed nature and ability to flow; often experience some type of intermolecular interaction; have a fixed volume but not a fixed shape
- Gas – the state of matter characterized by its non-condensed nature and ability to flow; molecules remain far apart from one another and show little interaction with other molecules; do not have a fixed volume or shape
- Plasma – ionized gas formed by superheating a gas; comprises over 99% of the visible universe
- Transitions between states:
- Melting – the transition from solid to liquid; the temperature increases until the substance reaches its meting point; as more heat is transferred, the substance begins to melt and become a liquid
- Freezing – the transition from liquid to solid; the temperature decreases until the substance reaches its meting point; as more heat is lost, the substance begins to freeze and become a solid
- Evaporation – the transition from liquid to gas; the temperature increases until the substance reaches its boiling point; as more heat is transferred, the substance begins to evaporate and become a gas
- Condensation – the transition from gas to liquid; the temperature decreases until the substance reaches its boiling point; as more heat is lost, the substances begins to condense and become a liquid
- Sublimation – the transition from solid to gas
- Deposition – the transition from gas to solid
3. Apply knowledge of the gas laws
- Gas laws – a set of observed relationships between a gas’s pressure, absolute temperature, volume, and amount
- Avogadro’s Law – the relationship between a gas’s volume (V) and amount (n, moles); at a constant temperature and pressure, a gas’s volume is directly proportional to its amount $$\frac{V_1}{n_1}=\frac{V_2}{n_2}$$
- Boyle’s Law – the relationship between a gas’s volume (V) and pressure (P); for a fixed amount of gas at a stable temperature, the gas’s volume is inversely proportional to its pressure $$P_1V_1=P_2V_2$$
- Charles’s Law – the relationship between a gas’s volume (V) and its temperature (T); for a fixed amount of gas at a constant pressure, the gas’s volume increases linearly as its absolute temperature increases $$\frac{V_1}{T_1}=\frac{V_2}{T_2}$$
- Gay-Lussac’s Law – the relationship between a gas’s pressure (P) and temperature (T); for a fixed amount of gas at a constant volume, the pressure of the gas is directly proportional to its absolute temperature $$\frac{P_1}{T_1}=\frac{P_2}{T_2}$$
- Ideal Gas Law – for a hypothetical ideal gas, the product of the pressure (P) and volume (V) of one gram of the gas is equal to the product of the absolute temperature (T), number of moles (n), and the gas constant (R); under constant temperature and pressure conditions, the volume of a gas depends on the amount (in moles) of the gas and not on the type of gas $$PV=nRT$$ $$\ gas\ constant: R=8.314\frac{m^3*Pa}{mol*K}$$
4. Identify the major discoveries in the development of the atomic theory
- Postulates of Dalton’s atomic theory (1807): solid sphere model
- Matter is composed of exceedingly small particles called atoms; an atom is the smallest unit of an element that can participate in a chemical change
- An element consists of only one type of atom, which has a mass that is characteristic of the element and is the same for all atoms of that element; a macroscopic sample of an element contains an incredibly large number of atoms, all of which have identical chemical properties
- Atoms of one element differ in properties from atoms of other elements
- A compound consists of atoms of two or more elements combined in a small, whole-number ratio; in a given compound, the numbers of atoms of each element is always present in the same ratio
- Atoms are neither created nor destroyed during a chemical change, but are instead rearranged to yield substances that are different from those present before the change
- J.J. Thomson (late 1800s): investigated the electrical discharges that could be produced by low-pressure gasses using a cathode ray tube (sealed glass tube with almost all air removed; contained two metal electrodes)
- When high voltage was applied across the electrodes, a visible beam (cathode ray) appeared between them and was deflected toward the positive charge and away from the negative charge; was produced in the same way with the same properties when the electrodes were composed of different metals
- The ray was simultaneously deflected by a magnetic field; measurements of the extent of the deflection and magnetic field strength allowed him to calculate the charge-to-mass ratio of the cathode ray particles; results indicated they were much lighter than atoms
- Thomson proposed that:
- The particles are attracted by positive (+) charges and repelled by negative (-) charges, so they must be negatively charged
- They are less massive than atoms and indistinguishable regardless of source material, so they must be fundamental, subatomic constituents of all atoms
- His cathode ray particle is what we now refer to as an electron
- Robert A. Millikan (1909): oil drop experiments
- Created microscopic oil droplets which could be electrically charged by friction as they formed or by using x-rays
- The droplets initially fell due to gravity; their downward progress could be slowed or reversed due to an electric field lower in the apparatus
- Was able to determine the charge on individual droplets by adjusting the strength of the electric field and making appropriate calculations
- The charge of an oil droplet is always a multiple of a specific charge $$\ 1.6*10^{-19}\,C$$
- Millikan concluded that this value must be a fundamental charge – the charge of a single electron
- The mass of a single electron was able to be calculated using the charge-to-mass ratio (Thomson) and the charge of an electron (Millikan) $$\ 9.107*10^{-31}\,kg$$
- Created microscopic oil droplets which could be electrically charged by friction as they formed or by using x-rays
- Scientists now established that the atom was not indivisible as Dalton had believed; the mass of the electron (-) was now known
- J.J. Thomson (1904): “plum pudding” model
- Described a positively charged mass with an equal amount of negative charge in the form of electrons embedded in it (since all atoms are electrically neutral)
- Hantaro Nagaoka (1903): postulated a Saturn-like atom consisting of a positively charged sphere surrounded by a halo of electrons
- Ernest Rutherford (1911): performed a series of experiments using a beam of high-speed positively charged alpha particles (a particles); a particles consist of two protons and two neutrons; produced by the radioactive decay of radium
- Rutherford, Geiger, and Marsden aimed a beam of a particles at a very thin piece of gold foil and examined the resulting scattering of the a particles using a luminescent screen that briefly glowed where hit by an a particle
- Most particles passed through the foil without being deflected
- Some particles were diverted slightly
- A very small number of the particles were deflected almost straight back toward the source
- Rutherford deduced:
- Because most of the a particles passed through the gold atoms undeflected, they must have traveled through virtually empty space
- Deflections arose when they encountered another positive charge
- Since the deflections only occurred a small fraction of the time, this charge only occupied a small amount of space
- Rutherford drew two conclusions:
- The volume occupied by an atom must consist of a large amount of empty space
- A small, relatively heavy, positively charged body (the nucleus), must be at the center of each atom
- Rutherford proposed a model in which an atom consists of a very small, positively charged nucleus, in which most of the mass of the atom is concentrated, surrounded by the negatively charged electrons, so that the atom is electrically neutral
- Rutherford also discovered that the nuclei of other elements contain the hydrogen nucleus as a “building block”
- He named this particle the proton
- Positively charged subatomic particle found in the nucleus
- He named this particle the proton
- Rutherford, Geiger, and Marsden aimed a beam of a particles at a very thin piece of gold foil and examined the resulting scattering of the a particles using a luminescent screen that briefly glowed where hit by an a particle
- Isotopes (early 1900s):
- Scientists identified several substances that appeared to be new elements after isolating them from radioactive ores
- A “new element” produced by the radioactive decay of thorium (initially named mesothorium) was shown to be chemically identical to radium, despite having a different atomic mass
- Led Frederick Soddy to realize that an element could have types of atoms with different masses that were chemically indistinguishable
- Called isotopes – atoms of the same element that differ in mass
- Was awarded the Nobel Prize in Chemistry in 1921 for this discovery
- Led Frederick Soddy to realize that an element could have types of atoms with different masses that were chemically indistinguishable
- A “new element” produced by the radioactive decay of thorium (initially named mesothorium) was shown to be chemically identical to radium, despite having a different atomic mass
- Scientists identified several substances that appeared to be new elements after isolating them from radioactive ores
- Niels Bohr (1913): planetary model; described the structure of atoms, particularly hydrogen
- First model that incorporated quantum theory; predecessor of quantum-mechanical models
- Describe the properties of atomic electrons in terms of a set of allowed possible values
- Atoms absorb or emit radiation when the electrons jump between allowed (stationary) states
- Direct evidence for the existence of such discrete states was obtained by Franck and Hertz (1914)
- Amended the view of the notion of the planetary electron model to bring the model in line with the spectral series of light emitted by hydrogen atoms
- By limiting the orbiting electrons to a series of circular orbits having discrete radii, Bohr could account for the series of discrete wavelengths in the emission spectrum of hydrogen
- Proposed that light only radiated from hydrogen atoms when an electron made a transition from an outer orbit to one closer to the nucleus
- The energy lost by the electron in the transition is precisely the same as the energy of the quantum of emitted light
- Proposed that light only radiated from hydrogen atoms when an electron made a transition from an outer orbit to one closer to the nucleus
- Electrons travel in defined circular orbits around the nucleus
- The orbits are labeled by an integer (the quantum number n)
- Electrons can jump from one orbit to another by emitting or absorbing energy
- By limiting the orbiting electrons to a series of circular orbits having discrete radii, Bohr could account for the series of discrete wavelengths in the emission spectrum of hydrogen
- Erwin Schrödinger (1926): quantum model
- Combined the equations for the behavior of waves with the de Broglie equation to generate a model for the distribution of electrons in an atom
- Assumes that the electron is a wave
- Tries to describe the regions in space (orbitals) where electrons are most likely to be found
- Describes the probability that an electron can be found in a given region of space at a given time; does not tell where the electron is, only where it might be
- Allowed the electron to occupy three-dimensional space; required three quantum numbers to describe the orbitals in which electrons can be found
- Principal (n), angular (l) and magnetic (m)
- Describe the size, shape, and orientation of the orbitals of the atom
- Combined the equations for the behavior of waves with the de Broglie equation to generate a model for the distribution of electrons in an atom
- James Chadwick (1932): found evidence of neutrons
- Neutrons – uncharged subatomic particles with a mass approximately the same as the mass of protons
- Explained the existence of isotopes
- Differ in mass because they have different numbers of neutrons, but are chemically identical because they have the same number of protons
- Explained the existence of isotopes
- Neutrons – uncharged subatomic particles with a mass approximately the same as the mass of protons
5. Identify the characteristics of elements, compounds, and mixtures
- Element – one of fewer than 118 pure chemical substances; composed of atoms with identical atomic number
- Made up of protons (+), electrons (-), and neutrons
- Protons (+) and neutrons are located in the nucleus of the atom
- Electrons (-) are located in orbitals surrounding the nucleus
- Have properties including:
- Conductivity – a measurement of a substance’s ability to transmit (conduct) heat, sound, or electricity
- Magnetism – forces of attraction or repulsion between objects
- Melting point – the temperature at which a substance is transformed from its solid state into its liquid state and where the solid and liquid states exist in equilibrium
- Boiling point – the temperature at which a substance is transformed from its liquid state into its gaseous state and where the liquid and gas states exist in equilibrium
- Color – a physical property of a substance relating to how light is absorbed and reflected
- State of matter – the physical state in which a substance exists
- Solid, liquid, gas, plasma
- Elements with similar properties are grouped together on the periodic table; where the element is positioned on the periodic table will determine how it reacts (or doesn’t react) with other elements
- Period – a row on the periodic table (1-7)
- Each period is associated with the number of energy levels that orbit a nucleus
- The number of electrons orbiting the nucleus will determine how that element interacts with other elements
- Group – a column on the periodic table (1-18)
- Have elements with identical valence electron counts and valence vacancy counts
- The similarity in composition and structure of their valence shells implies a corresponding similarity in both their chemical and physical properties
- Groups include:
- Alkali metals (1) – extremely reactive (easily lose electrons to become positive); generally found in nature combined with other elements; react with water to form alkalies (strong bases capable of neutralizing acids)
- Alkaline earth metals (2) – extremely reactive (easily lose electrons to become positive); typical compounds are ionic
- Halogens (17) – highly reactive ; free halogen elements are not found in nature; all produce sodium slats
- Noble gases (18) – colorless, odorless, tasteless, nonflammable gases; not very reactive
- Have elements with identical valence electron counts and valence vacancy counts
- Atomic number – the number of protons in an atomic nucleus
- Symbol – one or two letters representing a chemical element
- Atomic weight – the average mass of an atom of an element, usually expressed in atomic mass units; often used interchangeably with atomic weight
- Atomic mass unit (amu or u) – 1/12 the mass of the standard carbon-12 isotope $$\ 1.66*10^{-27} \,kg$$
- Period – a row on the periodic table (1-7)
- Made up of protons (+), electrons (-), and neutrons
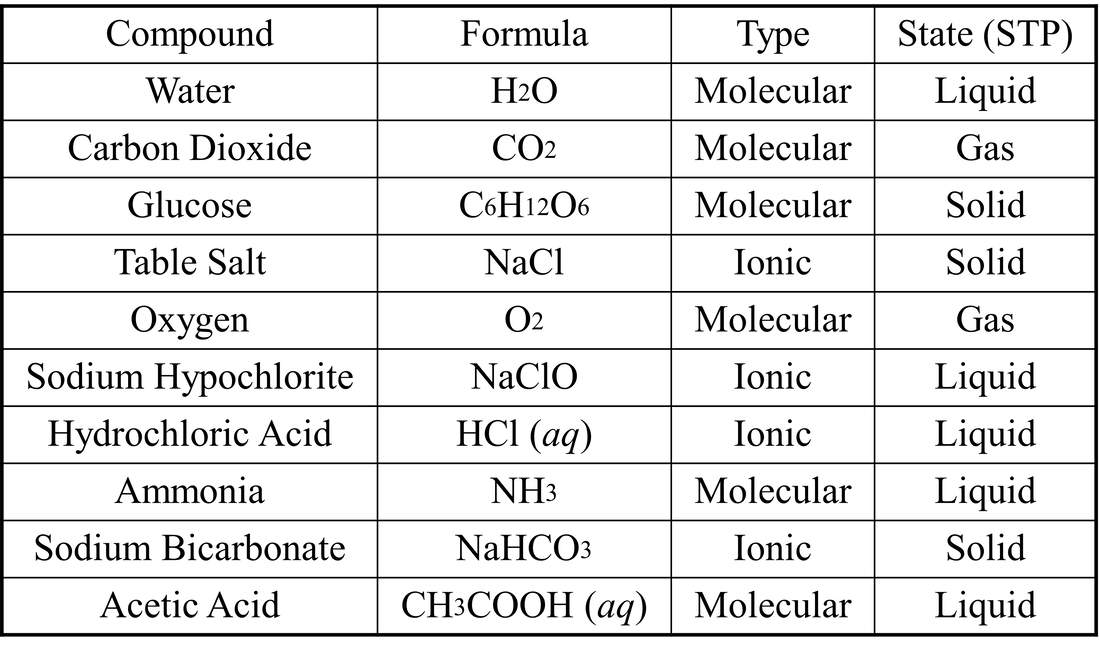
- Compound – a material formed by the chemical combination of elements in defined proportions; can be chemically decomposed into simpler substances
- Represented by a formula
- Indicates the types of atoms present with subscripts representing the relative numbers of atoms
- Ionic compounds – compounds held together with ionic bonds, an electrostatic attraction between positive (cations) and negative (anions) ions; commonly for complex lattices rather than true molecules
- Type of elements: metal and nonmetal
- Representative unit: formula unit
- Physical state at room temperature: solid
- Water solubility: usually high
- Melting and boiling temperatures: generally high
- Electrical conductivity: good when molten or in solution
- Molecular compounds – compounds held together by covalent bonds, formed by the bonding of two or more atoms; the smallest particle of a chemical compound that retains the chemical properties of the compound
- Type of elements: nonmetals only
- Representative unit: molecule
- Physical state at room temperature: gas, liquid, or solid
- Water solubility: variable
- Melting and boiling temperatures: generally low
- Electrical conductivity: poor
- Represented by a formula
- Mixture – two or more pure substances combined that each retain their unique properties; are physically combined and can be physically separated
- Homogeneous mixtures – also called solutions; cannot distinguish between the different parts
- Parts of a solution:
- Solute – a compound dissolved in a solvent to create a solution
- Solvent – the most abundant component in a homogeneous mixture of two or more substances; the substance in which a solute is dissolved
- Diluted solution – a solution with a small amount of solute
- Concentrated solution – a solution with a large amount of solute
- Solubility – the extent to which a substance dissolves in a particular solvent
- Parts of a solution:
- Heterogeneous mixtures – different parts can be distinguished from one another
- Homogeneous mixtures – also called solutions; cannot distinguish between the different parts
6. Apply knowledge of symbols, formulas, and equations for common elements and compounds (e.g. acids, bases, salts, carbon compounds) and their reactions
Common compounds:
- Acids – a substance that reacts with bases to form a salt; a compound that releases hydrogen ions (H+) in solution; a compound capable of donating hydrogen ions; a compound that can accept a pair of electrons from a base; indicated by a pH of less than 7
- Common acids:
- Hydrochloric acid (HCl)
- Sulfuric acid (H2SO4)
- Nitric acid (HNO3)
- Carbonic acid (H2CO3)
- Formic acid (HCOOH)
- Common acids:
- Bases – a substance that reacts with acids to form a salt; a compound that release hydroxide ions (OH-); a molecule or ion that accepts a pair of hydrogen ions from solution; a molecule or ion that donates an electron pair to an acid; indicated by a pH greater than 7
- Common bases:
- Sodium hydroxide (NaOH)
- Potassium Hydroxide (KOH)
- Magnesium Hydroxide (Mg(OH)2)
- Calcium Hydroxide (Ca(OH)2)
- Ammonia (NH3)
- Common bases:
- Salts – any ionic compound except those that contain hydroxide or hydrogen ions; any compound other than water formed by the reaction of an acid and a base
- Common salts:
- Sodium Chloride (NaCl) – simple salt
- Sodium Carbonate (NaHCO3) – acidic salt
- Ammonium chloride (NH4Cl) – acidic salt
- Potassium Cyanide (KCN) – basic salt
- Potassium Chlorate (KClO3) – neutral salt
- Sodium nitrate (NaNO3) – neutral salt
- Common salts:
- Carbon compounds – carbon is found free in nature in three forms: graphite, diamond, and fullerenes; all organic compounds contain carbon; forms compounds that make up ~18% of all the matter in living things
- Carbon Dioxide (CO2) – a greenhouse gas; dissolved in all natural waters
- Produced during cellular respiration
- Used during photosynthesis
- Carbon Monoxide (CO) – a dangerous asphyxiant; readily bonds to the hemoglobin in blood
- Hydrogen Cyanide (HCN) – extremely toxic protoplasmic poisons (inhibit tissue oxidation)
- Organic Compounds:
- Alkanes, Alkenes, and Alkynes
- Arenes – volatile liquids; include benzene and xylenes
- Alcohols and ethers – derivatives of water in which at least one hydrogen atom has been replaces by an organic group
- Aldehydes and Ketones – many have intense aromas
- Carboxylic acids – contain the carboxyl functional group
- Esters – have the general formula RCO2R’ where R and R’ can be virtually any alkyl or aryl group
- Amides
- Amines
- Carbon Dioxide (CO2) – a greenhouse gas; dissolved in all natural waters
7. Identify characteristics and functions of the components of an atom
- Parts of an atom:
- Nucleus – a tiny, dense positively charged mass at the center of an atom; composed of protons and neutrons and contains nearly al of the mass of the atom while occupying only a tiny fraction of the volume
- Protons charge: $$\ +1.60*10^{-19}\,C$$
- Proton mass: $$\ 1.672*10^{-27}\,kg$$
- Neutrons mass: $$\ 1.675*10^{-27}\,kg$$
- Can vary in number between atoms of the same element (isotopes)
- The number of protons commonly found in a particular element can be determined by subtracting the element’s atomic number from its atomic mass
- Electron shells – the orbitals around the nucleus of an atom where electrons reside; also called orbitals or energy levels
- Electron orbitals – three dimensional areas of space, defined by acceptable solutions to the Schrödinger equation, which determine the likely location of any given electron within an atom
- Assigned specific quantum numbers
- Principal quantum numbers (n=1, 2, 3, etc.)
- Orbital angular momentum quantum number (l)– can never be greater than n-1
- 0 = s; one s orbital; two s orbital electrons
- 1 = p; three p orbitals; six p orbital electrons
- 2 = d; five d orbitals; ten d orbital electrons
- 3 = f; seven f orbitals; 14 f orbital electrons
- Magnetic quantum number (ml) – can range from -l to +l
- Spin configurations (ms) – no two electrons can have the exact same quantum numbers, however, an electron can exist in spin up or spin down configurations
- The order of electron energy levels from least to greatest is: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p
- Principal quantum numbers (n=1, 2, 3, etc.)
- Assigned specific quantum numbers
- Electron charge: $$\ -1.60*10^{-19}\,C$$
- Electron mass: $$\ 9.11*10^{-31}\,kg$$
- Generally found around the nucleus of an atom
- May be gained or lost during ion formation
- Electron orbitals – three dimensional areas of space, defined by acceptable solutions to the Schrödinger equation, which determine the likely location of any given electron within an atom
- Nucleus – a tiny, dense positively charged mass at the center of an atom; composed of protons and neutrons and contains nearly al of the mass of the atom while occupying only a tiny fraction of the volume
8. Identify chemical or physical properties of elements based on their placement on the periodic table
- The periodic table – a table of chemical elements arranged in order of atomic number, usually in rows (periods), so that elements with similar atomic structure appear in vertical columns (groups)
- Metals – a material that shows a lustrous appearance when polished or fractured, and conducts electricity and heat relatively well; typically ductile and malleable as the result of metallic bonds
- Alkali metals – together with hydrogen, they form group 1 on the periodic table; have their outermost electron in the s orbital
- Lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr)
- Shiny, soft, highly reactive (at STP), readily lose an electron to form cations (+)
- Can be cut with a knife
- Shiny surface readily tarnishes due to atmospheric oxidation
- Must be stored under oil to prevent reaction with air
- Only found naturally as salts
- Alkaline earth metals – together with helium, they form group 2 on the periodic table
- Beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra)
- Shiny, silvery-white, somewhat reactive (at STP)
- Have relatively low densities, meting points, and boiling points
- React with halogens to form alkaline earth metal halides (ionic crystal compounds)
- React with water to form strongly alkaline hydroxides
- Lanthanides – often known as the rare-earth metals
- Actinides
- Transition metals – an element whose atom has a partially filled d sub-shell or which can give rise to cations with an incomplete d sub shell
- Post-transition metals – the metallic elements in the periodic table located between the transition metals and the chemically weak nonmetallic metalloids
- Gallium (Ga), indium (In), tin (Sn), thallium (Tl), lead (Pb), and bismuth (Bi)
- Alkali metals – together with hydrogen, they form group 1 on the periodic table; have their outermost electron in the s orbital
- Metalloids – a material that has properties that are in between or that are a mixture of those of metals and nonmetals
- Nonmetals – a material that generally lacks metallic properties; are usually poor conductors of heat and electricity; brittle or crumbly when solid; tend to attract electrons in chemical reactions; tend to form acidic compounds
- Reactive nonmetals –
- Halogens – group 17 on the periodic table
- Fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts)
- Free halogen elements are not found in nature due to their high reactivity
- Are all oxidizing agents
- Can combine with other elements to form halides
- Many halides can be considered salts of the respective hydrogen halides (colorless gases at STP) and form strong acids in aqueous solution
- Many halides can be considered salts of the respective hydrogen halides (colorless gases at STP) and form strong acids in aqueous solution
- Halogens – group 17 on the periodic table
- Noble gases – group 18 on the periodic table
- helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn)
- odorless, colorless monatomic gases with low chemical reactivity (at STP)
- Reactive nonmetals –
- Metals – a material that shows a lustrous appearance when polished or fractured, and conducts electricity and heat relatively well; typically ductile and malleable as the result of metallic bonds
9. Identify characteristics of types of chemical bonding (e.g. covalent, ionic, metallic, hydrogen)
- Covalent bond – a very strong chemical bond formed by the sharing of a pair of electrons
- Multiple covalent bonds can be formed when multiple pairs of electrons are shared between atoms
- Generally characterized as polar and nonpolar
- Polar covalent bond – a covalent bond in which electrons are unequally shared between bonding atoms
- Formed when one atom participating has a stronger affinity for electrons (electronegativity) than its bonding partner
- Will result in an unequal distribution of electron charge across the bonding pair
- Nonpolar covalent bond – a covalent bond in which electrons are equally shared between bonding atoms
- Formed when both atoms participating in a bond have an equal or nearly equal affinity for electrons (electronegativity)
- Will result in a symmetric distribution of electron charge across the bonding pair
- Polar covalent bond – a covalent bond in which electrons are unequally shared between bonding atoms
- Ionic bond – a chemical bond characterized by electrostatic attraction between ions of opposite charge
- The formation of an ionic bond involves a complete transfer of electrons between atoms and can be predicted when one bonding atom has a much higher electronegativity than the other
- Metallic bond – a bond between two metal atoms
- Strong and non-directional, meaning that it is hard to separate metal atoms but they can move and remain bonded as long as some parts of their surfaces are in contact
- The outer electrons are relatively free to move between bonded metal atoms
- Hydrogen bond – a strong dipole-dipole attraction between two or more molecules, at least one of which has a hydrogen atom bonded to an electron-withdrawing atom
- A weak bond formed between a hydrogen atom on one molecule which has developed a partial positive charge because of its bonding to an electronegative atom and an electronegative atom on another molecule
- A weak bond formed between a hydrogen atom on one molecule which has developed a partial positive charge because of its bonding to an electronegative atom and an electronegative atom on another molecule
10. Identify types of chemical reactions and their characteristics
- Chemical reaction – a process in which atoms and molecules recombine by forming or breaking chemical bonds; form new products that have different chemical properties than the initial reacting material
- Types of chemical reactions:
- Combination – a reaction in which two or more substances combine to form a single new substance; A + B → AB
- Example: 2Na (s) + Cl2 (g) → 2NaCl (s)
- Decomposition – a reaction in which a compound breaks down into two or more simpler substances; AB → A + B
- Example: 2HgO (s) → 2Hg (l) + O2 (g)
- Single-replacement – a reaction in which one element replaces a similar element in a compound; A + BC → AC + B
- Example: Mg (s) + Cu(NO3)2 (aq) → Mg(NO3)2 (aq) + Cu (s)
- Double-replacement – a reaction in which the positive and negative ions of two ionic compounds exchange places to form two new compounds; AB + CD → AD + CB
- Example: 2KI (aq) + Pb(NO3)2 (aq) → 2KNO3 (aq) + PbI2 (s)
- Combustion – a reaction in which a substance reacts with oxygen gas, releasing energy in the form of light and heat; must involve O2 as one reactant
- Example: 2H (g) + O2 (g) → 2H2O (g)
- Combination – a reaction in which two or more substances combine to form a single new substance; A + B → AB
- Stoichiometry – the proportional relationship (ratio) between reactants and products in a chemical equation